 Medtronic epidural stimulators first were used for spinal cord injury in 2009 under an Investigational Device Exemption with the FDA during research at UofL led by Susan Harkema, Ph.D., professor of neurological surgery and associate scientific director for KSCIRC. Angeli and Maxwell Boakye, M.D., neurosurgeon and clinical director of KSCIRC, will lead the project. Speed School of Engineering and director of the Epidural Stimulation Program at KSCIRC. Integrating multiple systems will allow people with chronic spinal cord injuries to benefit from stimulation on a daily basis by reducing the need to monitor and manually revise stimulation settings,” said Claudia Angeli, Ph.D., assistant professor of bioengineering in the UofL J.B. “We have seen excellent results with epidural stimulation in the lab, but these enhancements to the technology system will make it much easier to implement this therapy out in the community. The five-year project, funded through the NIH BRAIN Initiative, is focused on incorporating technology to improve control of locomotor and bladder function using epidural stimulation. The grant will fund work at UofL’s Kentucky Spinal Cord Injury Research Center ( KSCIRC) in collaboration with medical device manufacturer Medtronic to develop and test software applications specifically designed for spinal cord injury that work in concert with Medtronic’s commercially-available device, Intellis™, which is indicated as a spinal cord stimulator for chronic pain. The news gave hope to people living with complete spinal cord injuries, a diagnosis that historically meant they were unlikely to regain function below their level of injury.ĭespite these significant results, use of epidural stimulation outside a research lab setting to restore function for people with spinal cord injury thus far has been hampered by several limitations, including the use of a technology that was designed for patients with chronic, intractable pain – not those with spinal cord injury.Īpplying this therapy for spinal cord injury is a big step closer to use outside the research lab thanks to a $7.8-million grant from the National Institute of Neurological Disorders and Stroke, one of the National Institutes of Health (NIH). Researchers at the University of Louisville made news worldwide in 2018 when two people diagnosed with complete spinal cord injuries recovered the ability to walk thanks to experimental use of a therapy known as epidural stimulation. $7.8 million from NIH will fund development of a closed-loop system to monitor and adjust for multiple functions use, wireless monitoring UofL Health Movement Performance and Locomotor Recovery ClinicĪs originally published in the University of Louisville News.Activity-based Recovery Therapy Interventions.Recovery-Based Therapies: Clinical Program. 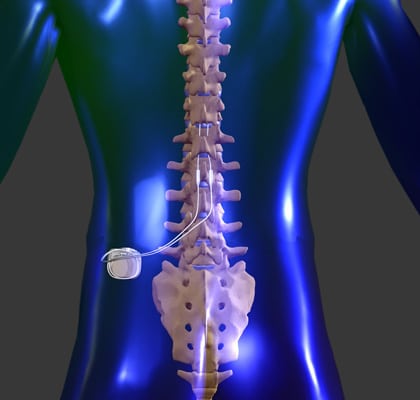
The Kosair for Kids Center for Pediatric NeuroRecovery.KSCIRC Biostatistics, Outcomes, Database, and Communications.Cardiovascular and Respiratory: Interventions.Cardiovascular and Respiratory: Inter-system Participation.Cardiovascular and Respiratory: Studies.Clinical and Community Fitness Partners. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
